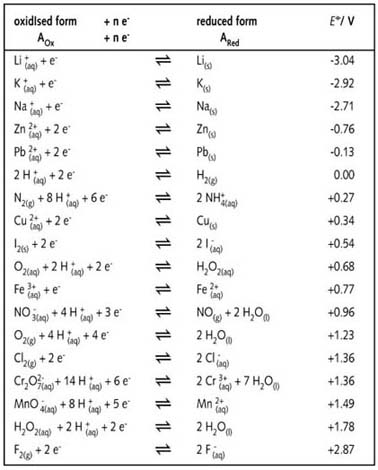
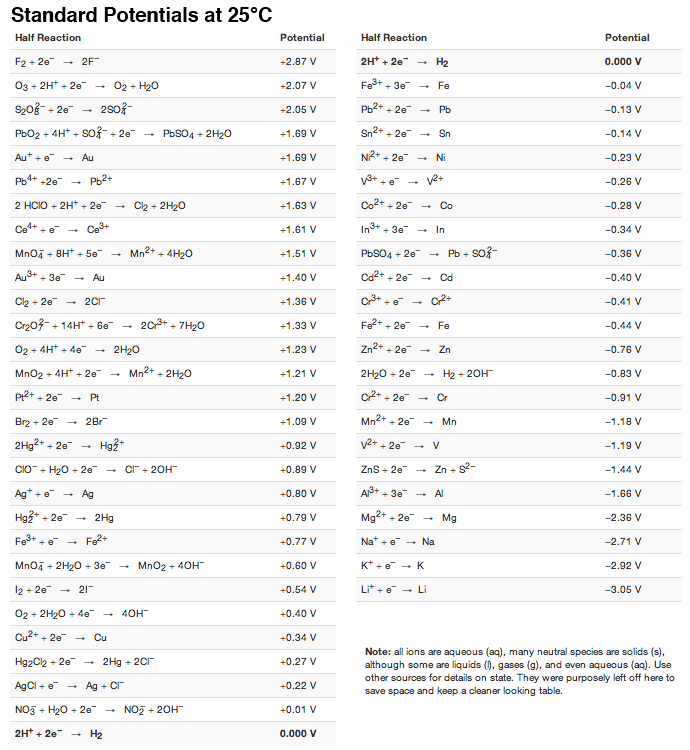
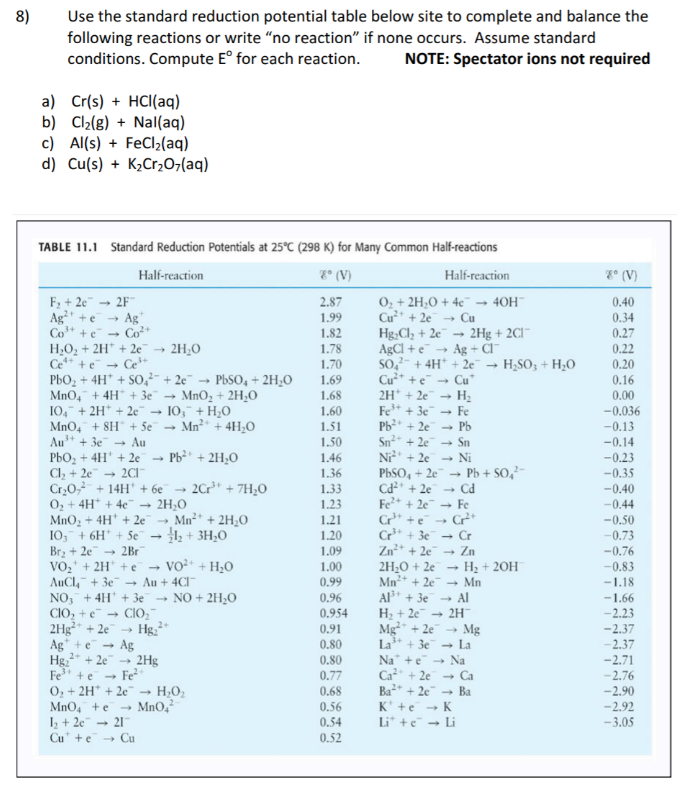
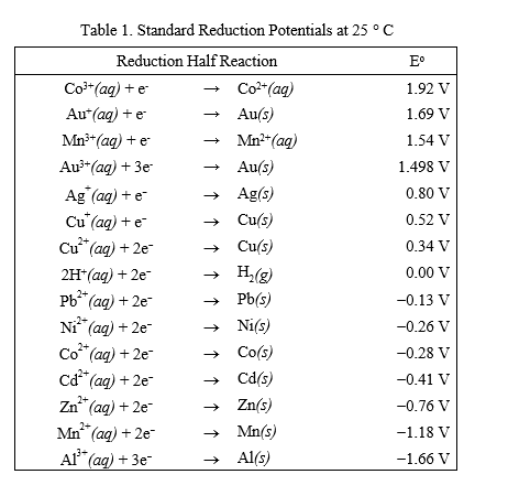
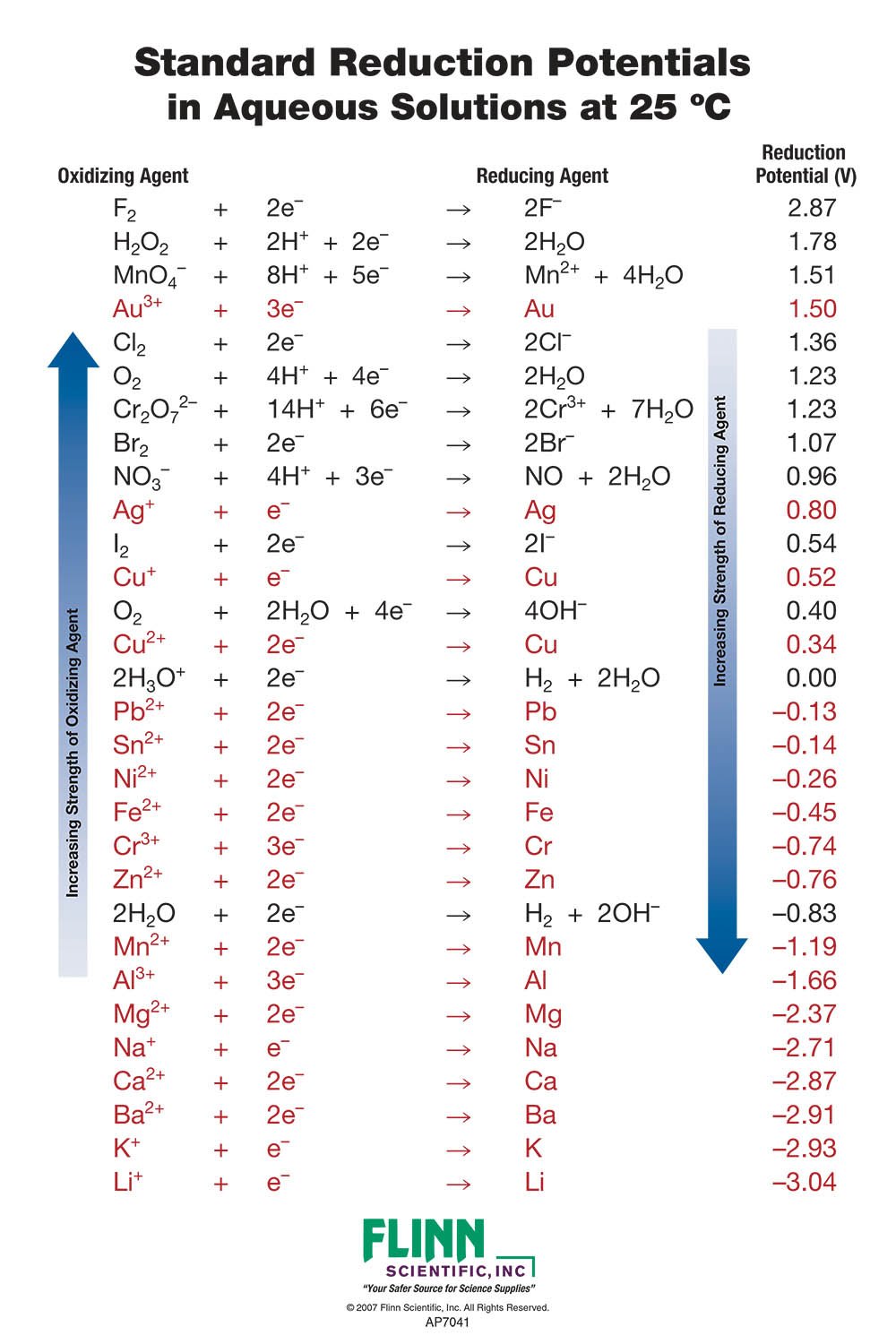
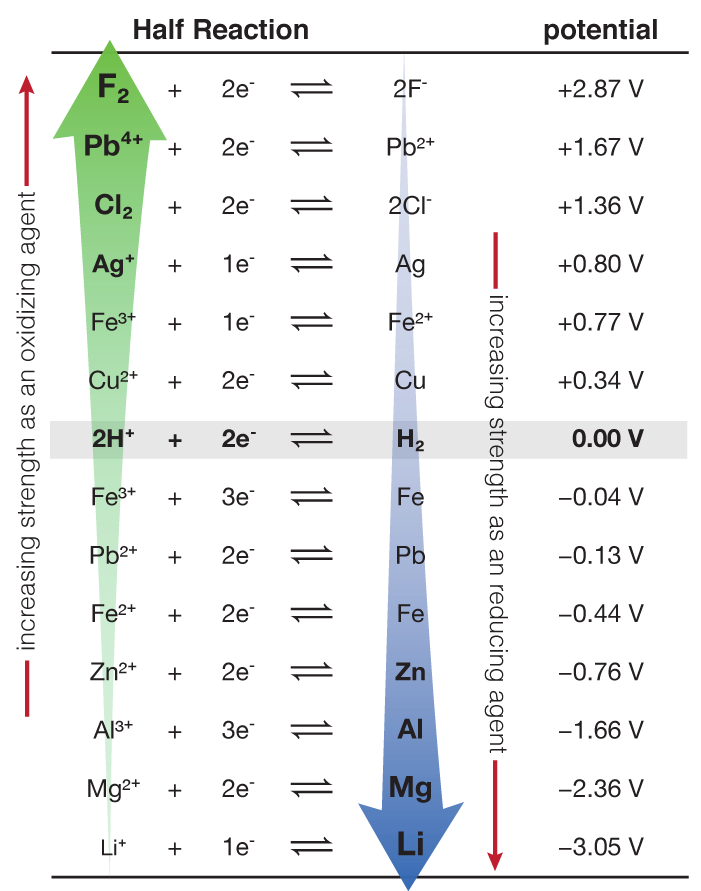
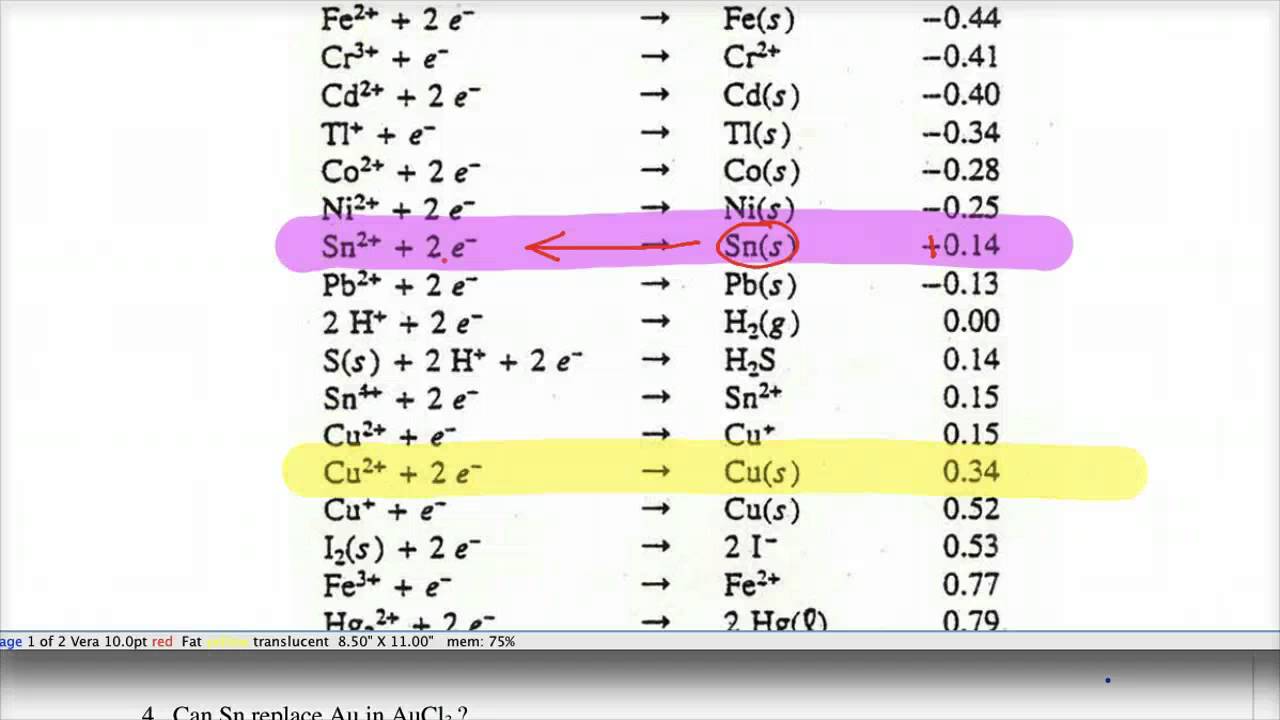
Using the standard electrode potentials given in the Table, predict the reaction between the following is feasible.Ag(s) and Fe^{3+}(aq)

Table 4 from Estimation of standard reduction potentials of halogen atoms and alkyl halides. | Semantic Scholar

Using the standard electrode potentials given in the table, predict the reaction between the following is possible.Fe^{3+}(aq) and I^{-}(aq)

Table 1 from Building and testing correlations for the estimation of one‐electron reduction potentials of a diverse set of organic molecules | Semantic Scholar

SOLVED: Table A5.5 Standard Reduction Potentials at 25°C (298 K) for Many Common Half-Reactions Half-Reaction Half-Reaction 4OH- + F2 â†' 2F- + 2H2O 2.87 2H2O + 4e- â†' Cu2+ + 2OH- 0.01

Table 1 from Absolute standard hydrogen electrode potential measured by reduction of aqueous nanodrops in the gas phase. | Semantic Scholar

WebElements Periodic Table » Periodicity » Reduction potential of hydrated M(I) ions » Periodic table gallery

Table of Standard reduction potentials.pdf - Table of Standard reduction potentials Half reaction Li e Li s K e K s Ca2 2e Ca s Na e | Course Hero